
Nanotubes crank out hydrogen
Pure hydrogen fuel is non-polluting. Current
methods of extracting hydrogen, however, use energy derived from sources
that pollute. Finding ways to use the sun's energy to split water to extract
hydrogen would make for a truly clean energy source.
Several research efforts are using materials engineered at the
molecular scale to tap the sun as an energy source to extract hydrogen
from water.
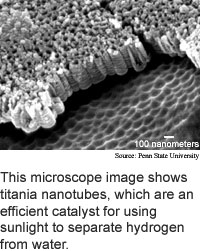
Researchers from Pennsylvania State University have constructed
a material made from titanium dioxide nanotubes that is 97 percent efficient
at harvesting the ultraviolet portion of the sun's light and 6.8 percent
efficient at extracting hydrogen from water.
The material is easy to make, inexpensive, and photochemically
stable, according to the researchers. The 97 percent efficiency is the
highest reported, according to the researchers. There is one catch --
only five percent of the sun's energy is ultraviolet light.
The researchers are working to find a way to shift the response
of the nanotube arrays into the visible spectrum.
The key to making titanium dioxide nanotubes that efficiently
harvest the energy from light is controlling the thickness of the nanotube
walls, according to the researchers. Nanotubes 224 nanometers long with
34-nanometer-thick walls are three times more efficient than those that
are 120 nanometers long with 9-nanometer-thick walls.
The researchers made the titanium dioxide nanotube material by
mixing titanium with acid and electrifying the mixture, which caused the
tiny tubes to grow, then heating them to cause the material to crystallize.
The material could be ready for practical use in two to five years,
according to the researchers. The work appeared in the January 12, 2005
issue of Nano Letters.
Atomic scopes eye living tissue
Brainwave interface goes 2D
Software ties marks to digital text
View from the High Ground:
Xerox's Hervé Gallaire
Briefs:
All-silicon chip laser demoed
Nanotubes crank out hydrogen
Nanotubes on plastic speed circuits
Lens design promises tight spots
Silicon nanocrystal transistor shines
Nano triangles concentrate light
Musical additions sound alarm
Research Watch blog
View from the High Ground Q&A
How It Works
RSS Feeds:
News
Ad links:
Buy an ad link
Ad links: Clear History
Buy an ad link
|
TRN
Newswire and Headline Feeds for Web sites
|
© Copyright Technology Research News, LLC 2000-2010. All rights reserved.